Anti-Neoplastic Effects of Calcium in Colorectal Cancer Potentiated by Active Vitamin D
Febuary, 2015
Groups from Vienna published a paper in 'The Journal of Steroid Biochemistry and Molecular Biology' about Vitamin D potentiating the anti-neoplastic effects of calcium in the colon.
Introduction
Colorectal cancer (CRC) is the third major cause of death in almost all regions of the world. However, it is among the most slow-growing cancers of the world, with its roots lying in improper diet in 70-90% of cases. This means it is among the most preventable of cancers. Both epidemiological and clinical studies are supported by experimental evidence that calcium and vitamin levels in the diet are inversely proportional to the risk for CRC, though this evidence is not perfect.
As a result, the Second Expert Report, jointly published by the World Cancer Research Fund and the American Institute for Cancer Research, lists calcium as one of the factors that are most likely responsible for a lowered risk of the disease, but vitamin D has limited supportive evidence, which is nonetheless suggestive of a protective effect.
Studies of colonic mucosa suggest that there is a normal balance between cellular proliferation, differentiation and apoptosis of the mucosal cells which protects against neoplastic proliferation. When this is upset, there is an increased risk of CRC.
This balance is due in part to calcium and vitamin D. The lack of evidence in clinical studies has led to an increased interest in combination chemoprophylaxis based on the concept of a cross talk between calcium and vitamin D molecules. These signals have a protective effect on the development of CRC.
The impetus for this research came from three trials which showed a favorable effect of combined calcium and vitamin D on CRC. A randomized placebo-controlled clinical study by Grau et al showed that the combined administration of calcium and vitamin D brought down the CRC risk.
Holt et al also conducted a trial which demonstrated lower rates of polyp formation in adenoma patients who were on combined calcium and vitamin D supplementation. Mortality from CRC was also lower in patients with high intakes of dietary calcium (928 mg/d or more) and elevated serum 25-hydroxyvitamin D levels.
Vitamin D3 is metabolized by hepatic hydroxylation at the C-25 position to 25-hydroxyvitamin D3 (25-D3) by the enzymes vitamin D 25-hydroxylases CYP2R1 and CYP27A1. This is then bound to vitamin D binding protein (DBP) and transported to the kidney for another hydroxylation at C-1.
This is catalyzed by the enzyme 25-D3 1α-hydroxylase (CYP27B1) and the end-product is vitamin D, 1,25-dihydroxyvitamin D3 (1,25-D3), which is the biologically active form. To smaller extents, 1,25-D3 is produced and broken down at other sites in the body, including the intestine.
How is calcium related to vitamin D? The calcium-sensing receptor, CaSR, is a G-protein coupled receptor whose activity is enhanced by a promoter element that contains two vitamin D response elements (VDRE). In response to vitamin D the CaSR is upregulated, and thus calcium tissue effects are increased.
These effects include calcium homeostasis and cell growth. The latter is key to the present discussion, since one study involving CaSR knockout mice showed hyper-proliferation of the cells lining the colonic crypts and a higher vulnerability to the formation of areas of aberrant crypts.
Loss of CaSR expression is characteristic of advanced undifferentiated tumors. The current experiment is therefore aimed at gathering data to elucidate the role of the CaSR in preventing neoplasia of the colon. It was carried out to assess the extent to which the level of CaSR expression and function affect vitamin D signaling and enhance calcium effects within the colonic mucosa.
Immunohistochemistry
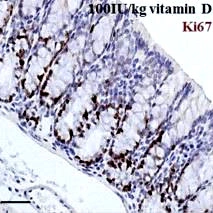
In the present experiment, 4 μm sections were taken and Ki67 stain applied (1:400, Novus Biologicals, USA) with hematoxylin (DAKO, Austria) as counter stain, on a Ventana Discovery XT autostainer (Ventana, USA), after pretreating with cell conditioner 1. The detection system used was the rabbit OmniMap (Ventana). The automated TissueFAXS PLUS system (TissueGnostics, Austria) was used to acquire whole section images.
To evaluate the sections, at least 10 intact crypts per colon were identified and the percentage of Ki67-positive cells per crypt was quantified in a blinded fashion. Care was taken to include both the ascending and descending colon.
Discussion
While research seems to indicate a role for calcium and vitamin D in preventing, modifying, or reversing neoplastic changes in colonic mucosa, the current experiment is meant to show the effect of a diet with high vitamin D content on proliferation (reduced), differentiation (increased) and apoptosis (increased), along with simultaneous upregulation of the expression of CaSR in the mouse colon.
This mechanistic model may show that molecular cross talk at the level of the calcium and vitamin D occurs directly in the colon via the CaSR, and that this is central to the anti-neoplastic effects of this combination. The gene expression and functionality of CaSR is proportional in turn to the preventive impact of active 1,25-D3 on CRC.
Prior experiments demonstrated a downregulation of the vitamin D catabolic enzyme1,25-dihydroxyvitamin D3 24-hydroxylase (CYP24A1) in the colon of mice who had higher dietary intakes of calcium. On the other hand, a low calcium intake caused colonic crypts to show increased proliferation and low apoptosis rates.
The current experiment was performed in vivo and demonstrated that a high vitamin D intake had much higher CaSR expression, decreased proliferation, and increased differentiation and apoptosis in normal colonic mucosa. The conclusion therefore was that CaSR could well link the cross talk between calcium and vitamin D, which means that their efficiency as combined chemoprophylaxis of CRC depends upon its activation and regulation by this receptor.


